 It is also used in medicine, in electroplating, and in agriculture.Ĭobalt(II) fluoride, a compound with the formula CoF2, is used as a catalyst to alloy metals. Cobalt can be used in the production of catalysts too. Cobalt had been used in imparting a blue color to glazes, glass, and ceramics. 
Cobalt compounds are used in the manufacture of porcelain enamels and batteries as well as in paints and inks. Cobalt blue has been used as a coloring agent in jewelry and Chinese porcelain. It also alloys with aluminum, platinum, iron, and nickel. Uses of CobaltĬobalt, a chemical element with the symbol Co, is used in making magnets. Due to its high strengths, the alloys of this metal are used in aircraft production, etc. With industrial purposes, cobalt is usually used in alloys with iron, nickel, and aluminum, to produce powerful magnets. If consumed in larger doses, cobalt is very carcinogenic. This element is crucial for all living organisms, but in micro doses. This element can be found in some minerals, as it was discovered that there are some cobalt deposits at the bottom of the oceans. This metal used to be one of the materials widely used in ancient China to produce porcelain and add a bit of beautiful blue color to it. The metal is rarely used as a structural material in the pure form but almost always as an alloy or a component of another system. Its name is believed to take roots from a German word meaning goblin. Cobalt boride is produced under high temperature such as 1500 ☌. Most Western countries have their supplies of cobalt imported, as it is primarily mined in Africa.Cobalt is a light grey and bluish metal, which is similar to iron in its qualities and can be easily magnetized. However, there are also small traces of cobalt that can be found in some types of fertilizers. This means that it was discovered after modern language was invented, so that the information (such as the name of the discoverer) could be written down.Ĭompound cobalt-60 is used in the production of radioactive emissions used in chemotherapy to help treat cancer and used in the cleaning and sterilizing process of medical tools. It was the first metal to be discovered that occurred recently enough in history that we know the name of the person that discovered it. More facts about cobaltĬobalt was the first metal to be discovered after the development of civilization, after the prehistoric era. Mined cobalt is used as a dye for blue coloring in various ink, paint, ceramic and glasswork, and sometimes cosmetic products.Īncient Chinese and Roman glass- and ceramic-making techniques often used to include compounds of cobalt.īecause of cobalt’s magnetic properties, it can also be used in the creation and production of batteries, magnets, and other industrial equipment. Most of cobalt mining is done on the African continent, and these mining efforts tend to include the mining of nickel, iron, copper, lead, and silver as well. It is not generally found freely around the planet Earth, but it can be mined from various other minerals and rock from the Earth’s outer crust. The human body, along with other animal bodies, use naturally-occurring cobalt to create important chemicals in the body, such as enzymes.Ĭobalt is one component of the well-known, important vitamin B12.Ĭobalt was discovered by George Brandt in the year 1735. What is the melting point of cobalta)1449 Kb)1559 Kc)1669 Kd)1769 KCorrect answer is option D. Interestingly, cobalt is also important in the survival of living organisms. Quick links for Chemical Engineering exam. Cobalt is best known as a raw material in lithium-ion batteries, but its high melting point and ability to maintain strength even at raised temperatures make it useful in cutting tools. In general, cobalt is resistant to corrosion, rust, and other damage.  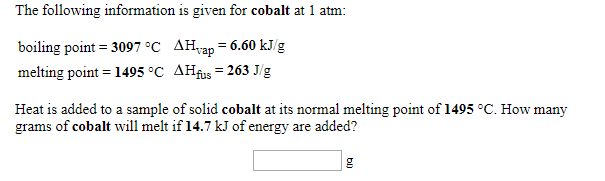
Cobalt primarily appears as a hard, breakable metal that looks blue and white in color.Ĭobalt’s atomic number is 27, of course, and its atomic weight is 58.93.Īt room temperature, cobalt is a solid, and its melting point is very high at 1495 degrees Celsius.Ĭobalt also has magnetic properties, and continues retain these magnetic properties even when heated to very high temperatures.Ĭobalt is only very minorly reactive, and any reaction that it has from being exposed to oxygen occurs very slowly.Ĭompounds can be formed in the combinations of cobalt and other elements, leading to cobalt oxide, cobalt fluoride, and cobalt sulfide.īecause the melting point of cobalt is so high and has little to no reaction to oxygen, it is considered to be very stable at even very high temperatures. Atoms of cobalt are known to have 27 protons, 27 electrons, and 32 neutrons. Cobalt is an element from the periodic table of elements that falls into the ninth column of the table.Ĭobalt is considered a transition metal due to its location on the periodic table and some of its properties as a metal. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
